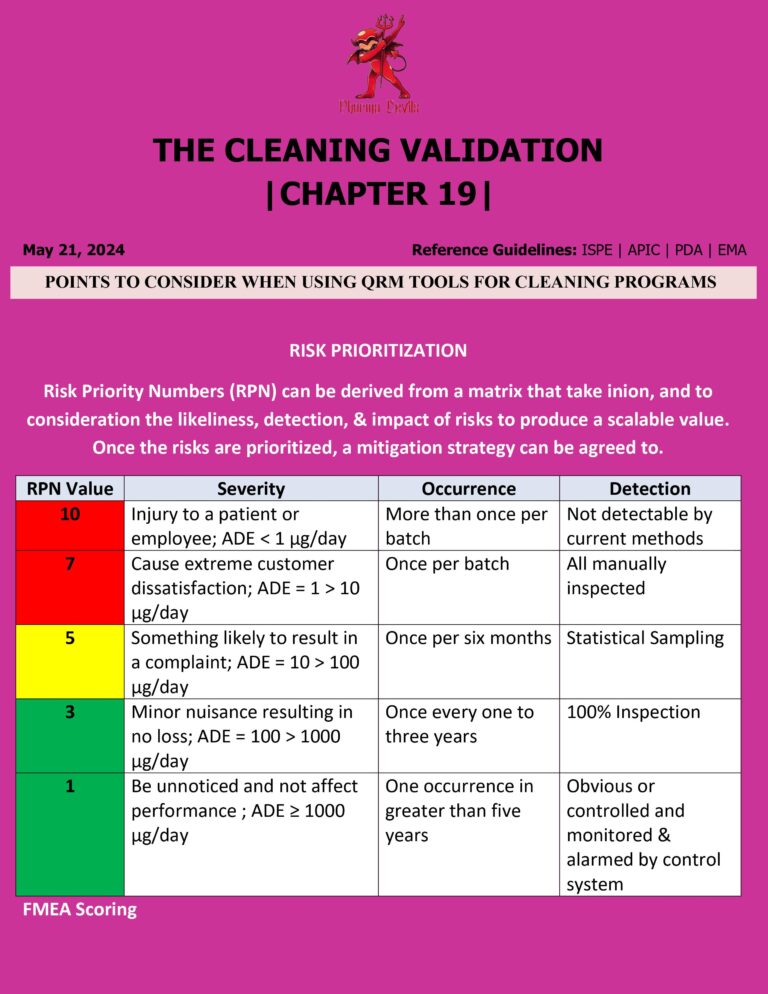
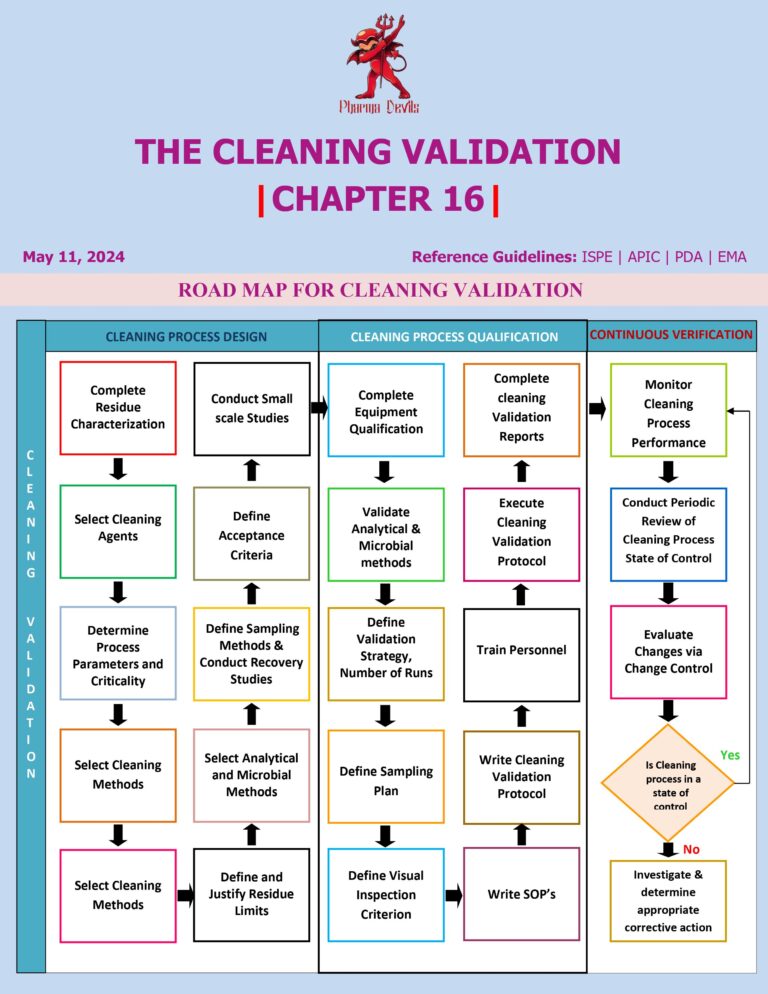
LYOPHILIZER QUALIFICATION IN PHARMA

Lyophilizer Qualification in Pharma is an important aspect in pharma. Lyophilization, also known as freeze-drying, is a process used to preserve a perishable material or make it more convenient for transport. It works by freezing the material and then reducing…