SOP FOR ANALYTICAL, RESEARCH AND DEVELOPMENT IN PHARMA

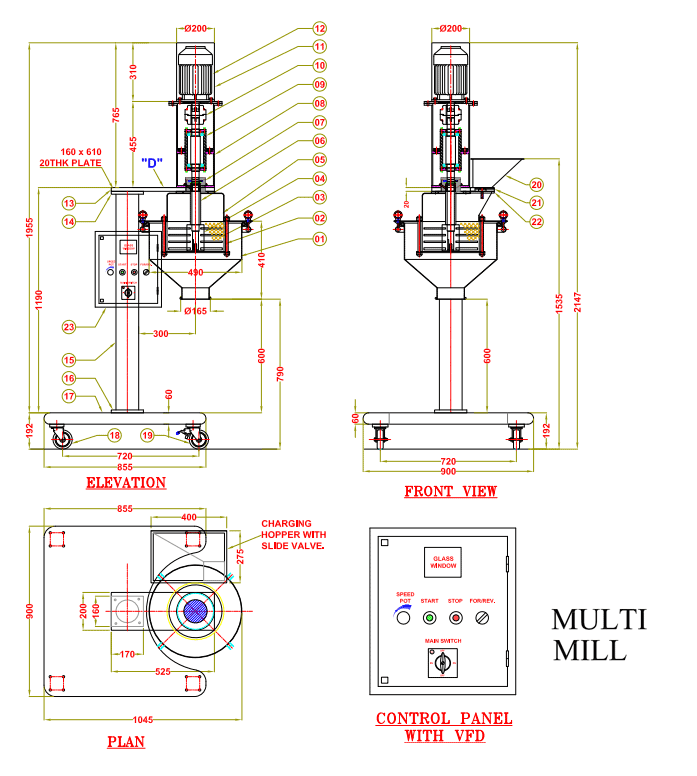
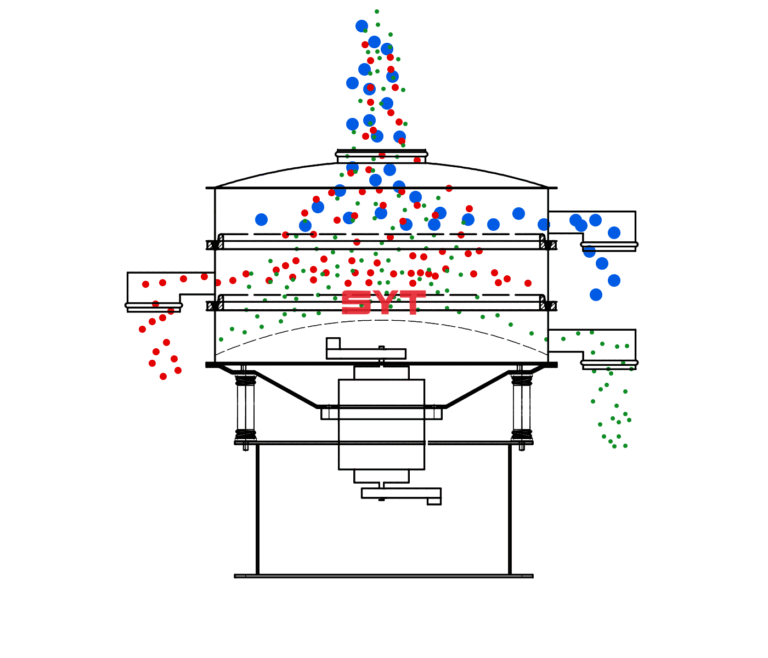
Analytical Research & Development (Analytical R&D) in pharma is the function that develops, validates, and transfers test methods needed to characterize materials and ensure drug products meet quality requirements throughout development and commercialization. It bridges discovery, formulation/process development, manufacturing, QC,…