PURIFIED WATER PRE-TREATMENT, GENERATION, STORAGE & DISTRIBUTION SYSTEM
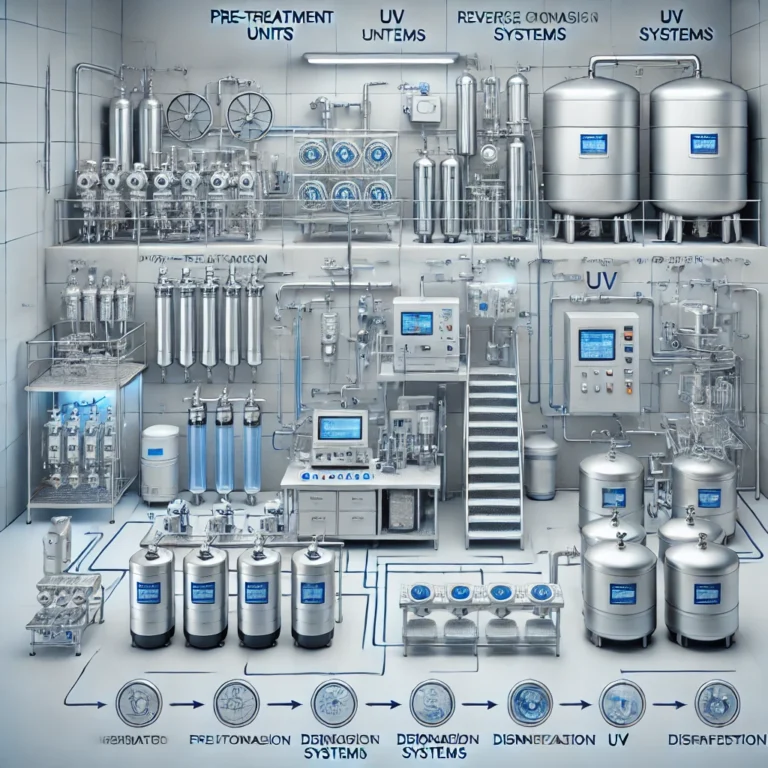
Here’s a practical, end-to-end overview of a Purified Water (PW) system in pharma: pre-treatment → generation → storage → distribution—what each stage does, typical equipment, and key GMP controls. 1) Pre-treatment (protect the RO/EDI and stabilize feed quality) Purpose: reduce…














