Risk Assessment for APQR
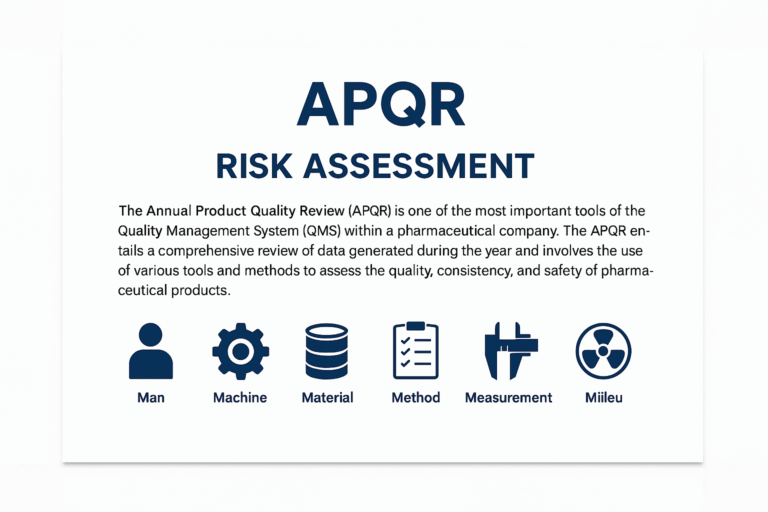
Introduction The Annual Product Quality Review (APQR) is a critical quality management activity mandated by global regulatory authorities including the US FDA, EU GMP (Annex 15 & 16), WHO, and ICH Q10. The purpose of the APQR is to systematically…














