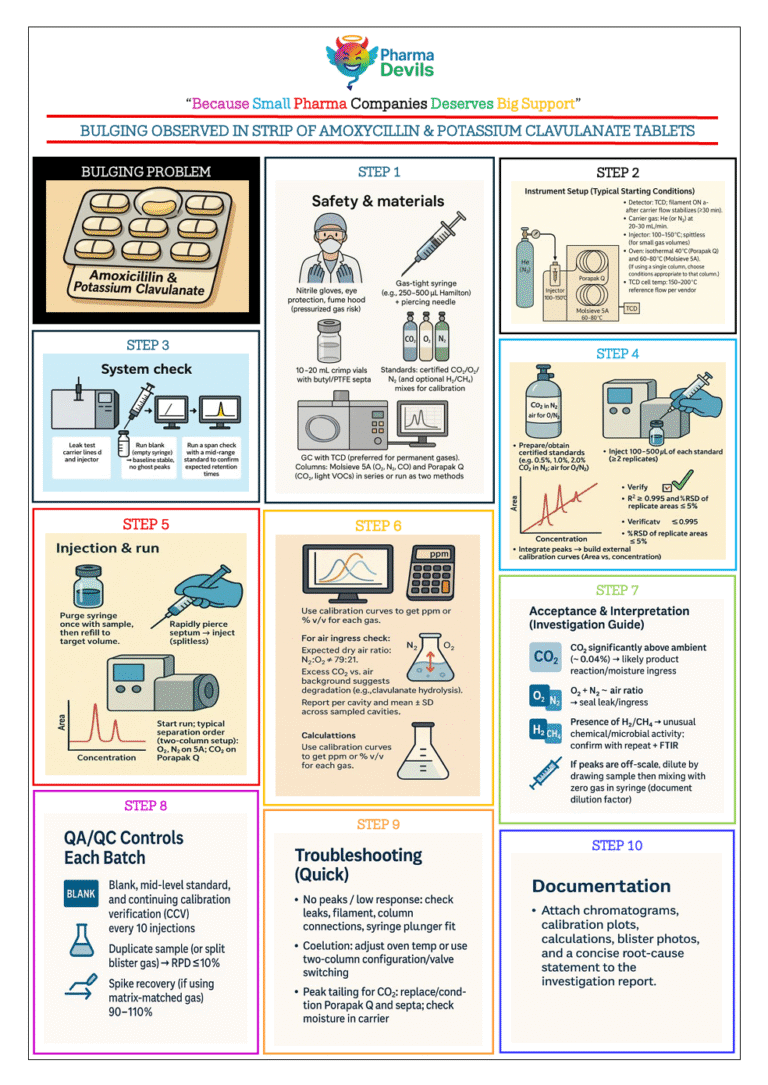
ENTRY PROCEDURE IN MANUFACTURING CORRIDOR [PICTORIAL]

1.0 OBJECTIVE To define the standard entry procedure for personnel into the manufacturing corridor of the oral solid dosage (OSD) area to prevent contamination, mix-ups and to maintain GMP. 2.0 SCOPE This procedure is applicable to all personnel (Production, QA,…