














AHU qualification in pharma is a documented verification process used to ensure that the Air Handling Unit performs as per approved design, GMP requirements, and cleanroom environmental standards. AHU plays a critical role in pharmaceutical manufacturing by controlling airflow, temperature, relative humidity, pressure differential, air filtration, air changes, and contamination control.
The purpose of AHU qualification is to confirm that the system is properly installed, operates correctly, and consistently maintains required environmental conditions in production, packing, sampling, dispensing, microbiology, and other controlled areas. AHU qualification is generally performed during new installation, modification, relocation, major maintenance, filter replacement, or periodic requalification.
The qualification process includes Design Qualification, Installation Qualification, Operational Qualification, and Performance Qualification. During Design Qualification, the AHU design is reviewed for capacity, airflow pattern, filter selection, ducting, dampers, sensors, cooling coil, heating coil, humidification, dehumidification, and pressure control. Installation Qualification verifies that the AHU, ducts, filters, gauges, instruments, and utilities are installed according to approved drawings and specifications.
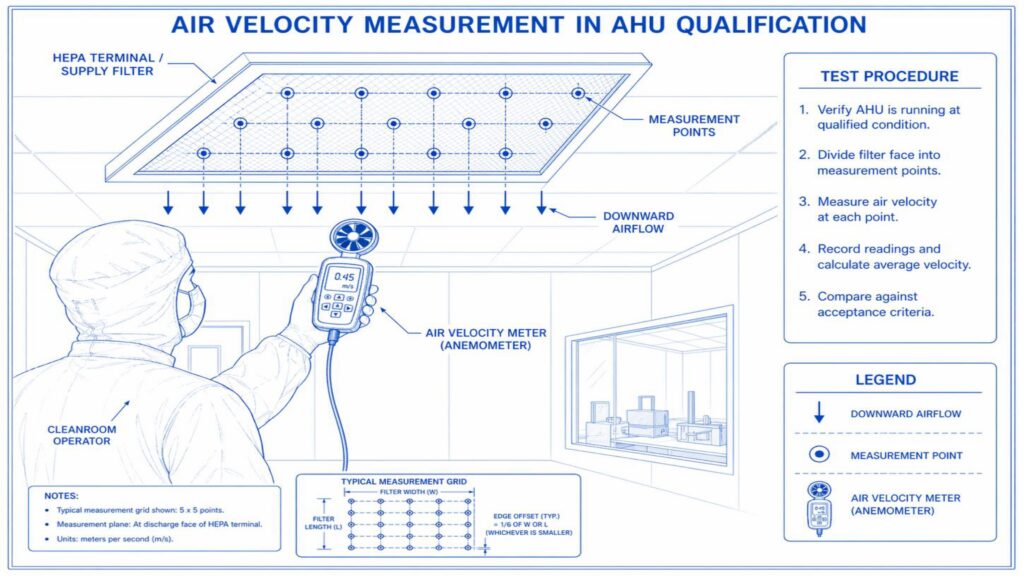
Operational Qualification confirms that the AHU operates within defined limits. Tests may include blower operation, damper function, filter integrity, alarm verification, interlock checks, temperature control, humidity control, differential pressure monitoring, and air velocity measurement. Instruments used for testing must be calibrated.
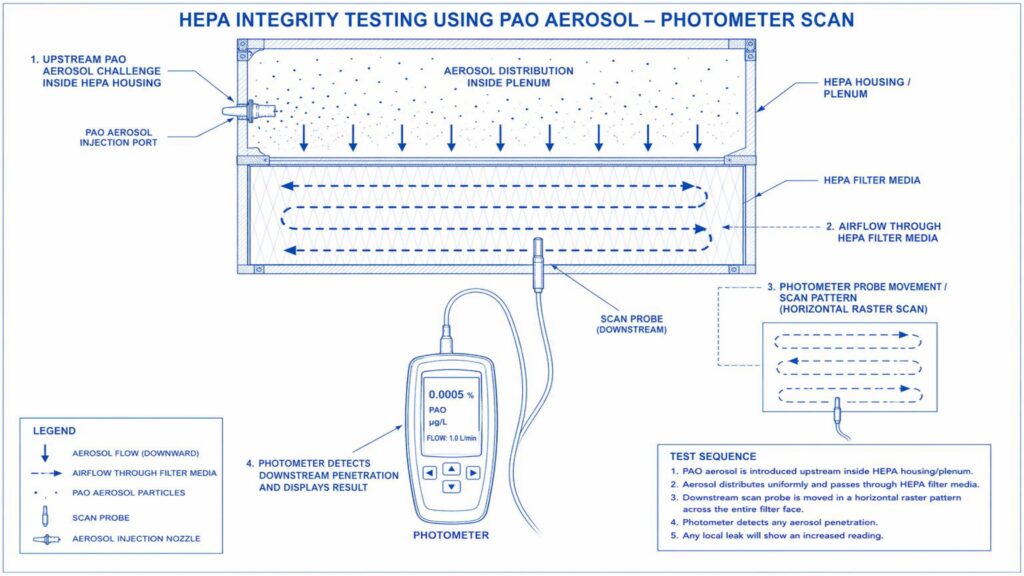
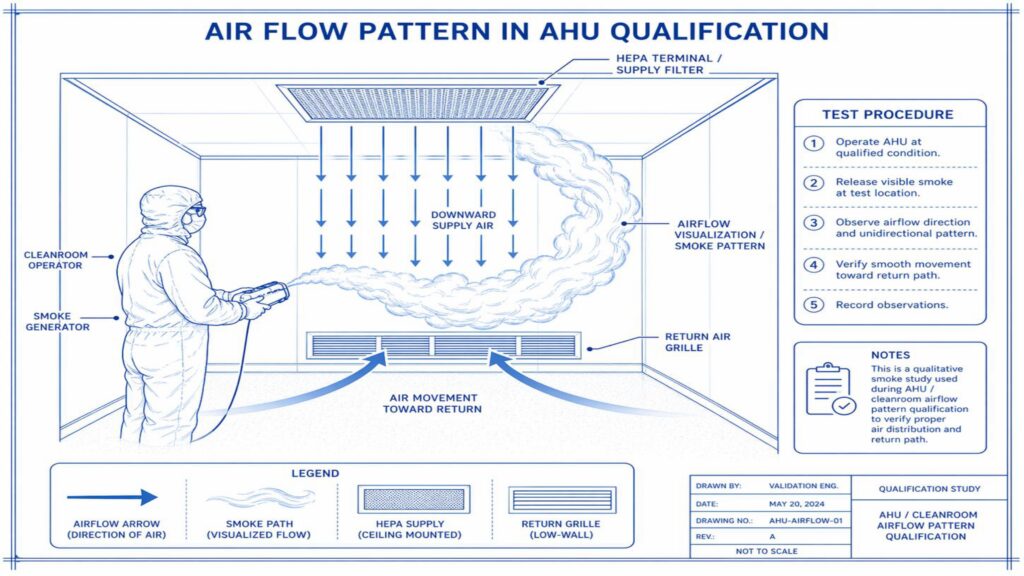
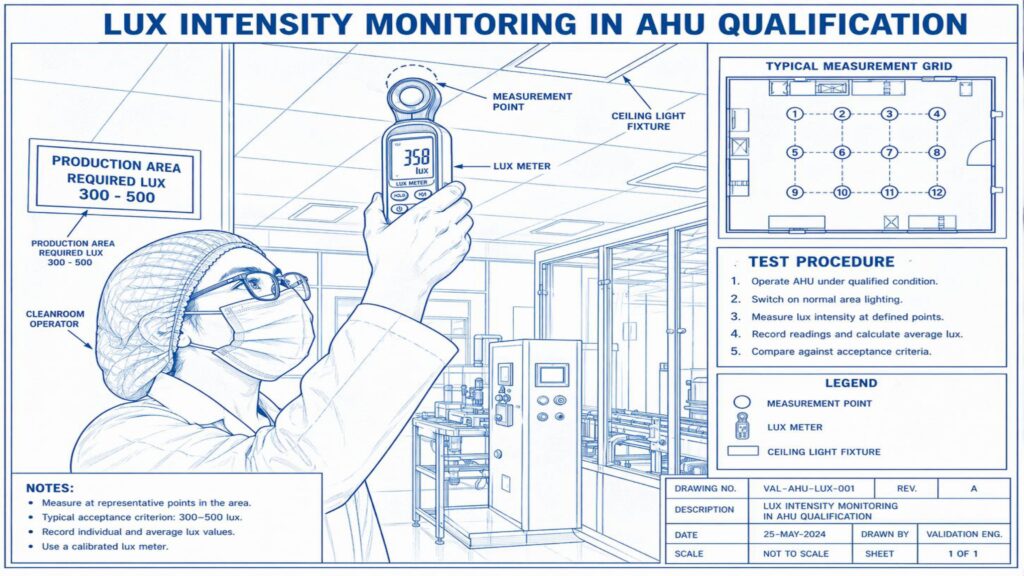
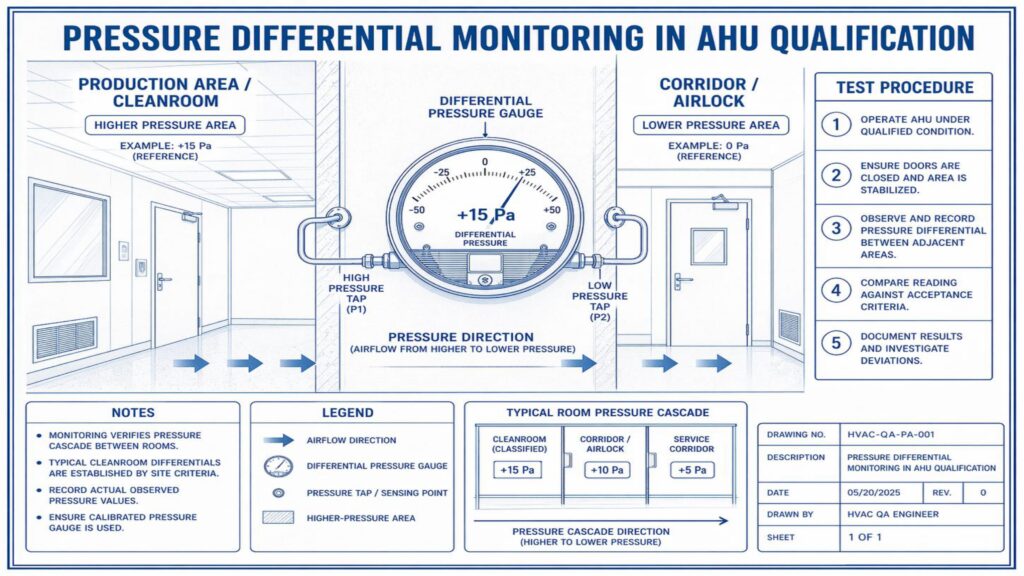
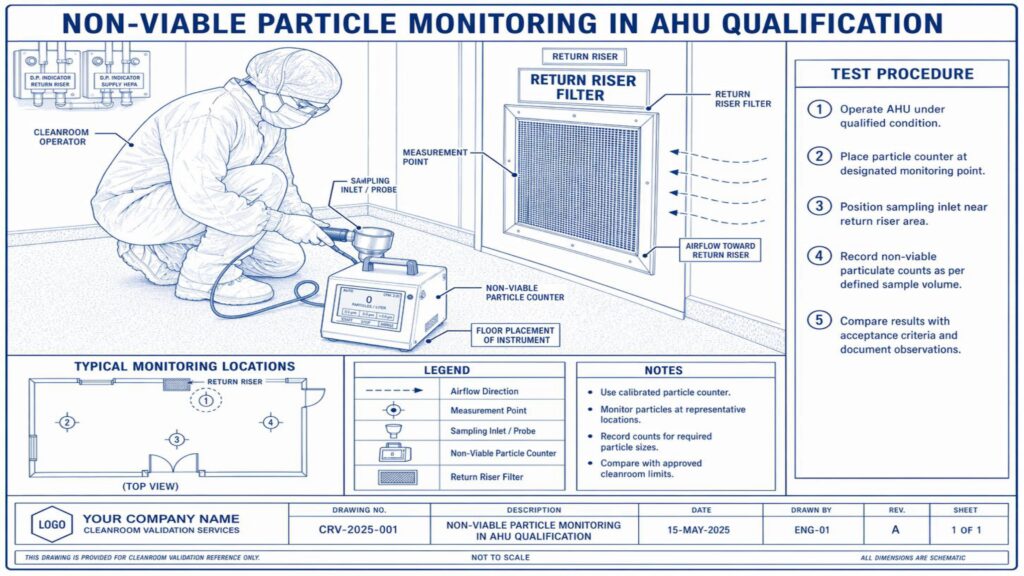
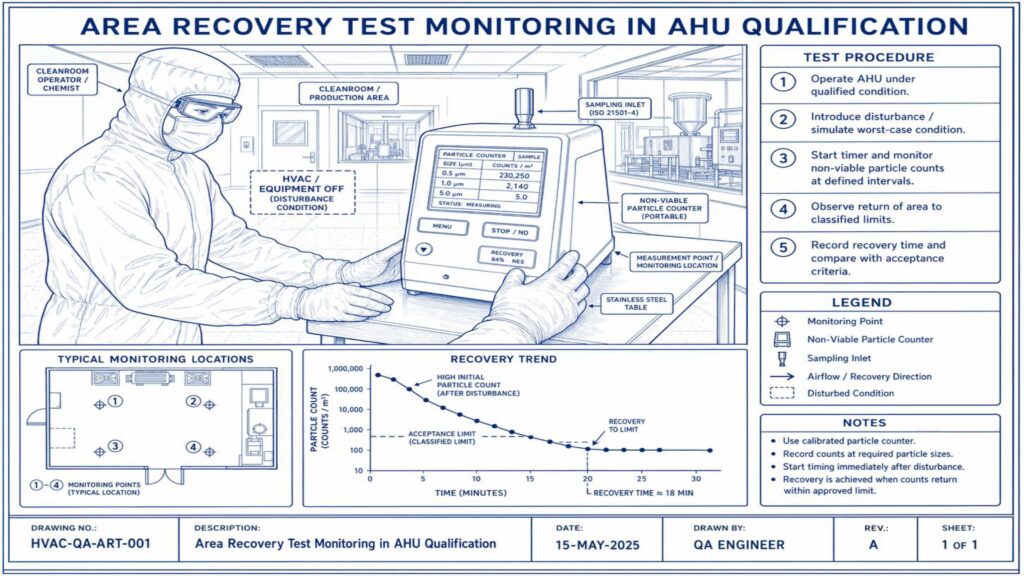
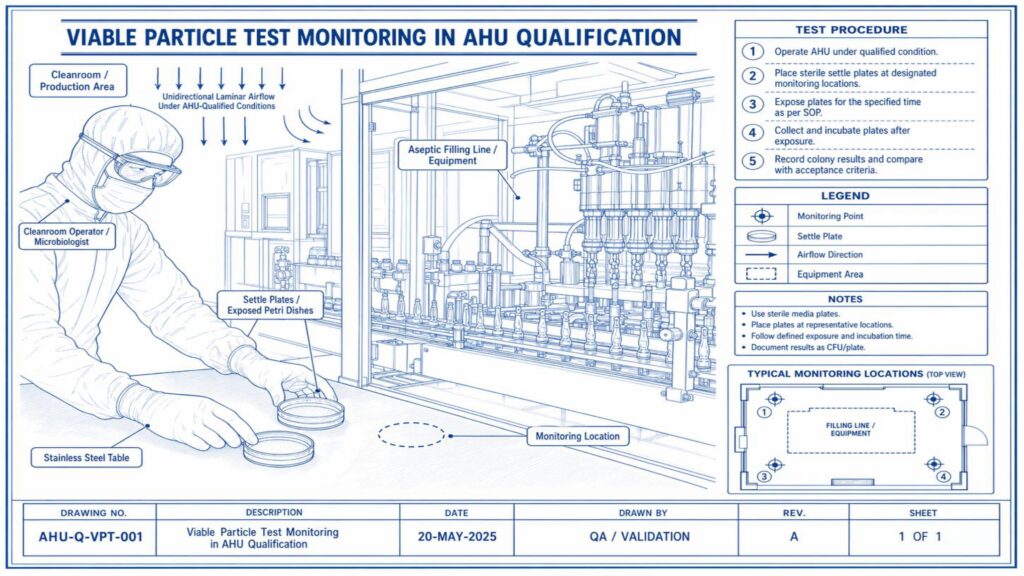
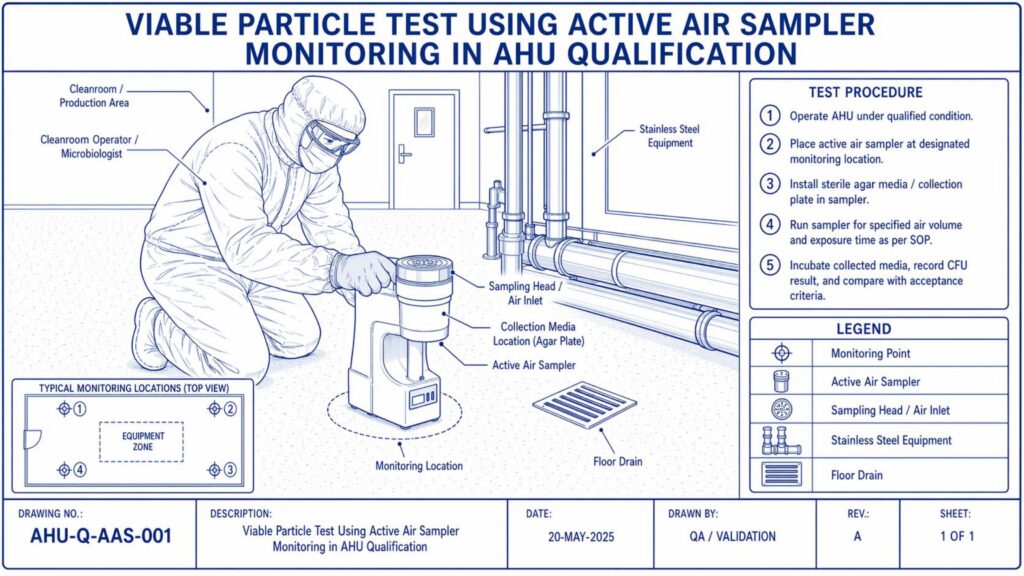
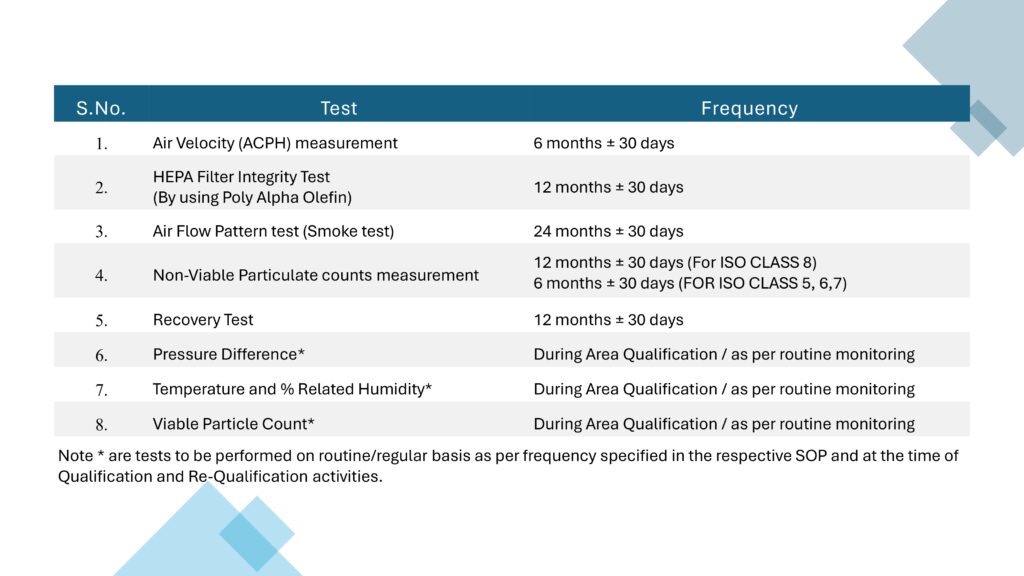
Performance Qualification verifies the actual performance of the AHU under routine operating conditions. Important tests include air change rate, airflow volume, room pressure differential, temperature and humidity mapping, HEPA filter leak test, non-viable particle count, recovery test, smoke study, and microbial monitoring where applicable.
Acceptance criteria should be predefined in the approved qualification protocol. Any deviation, abnormal result, or failure must be investigated, corrected, and documented before approval. Final qualification reports should include test results, observations, calibration details, deviations, corrective actions, and QA approval.
A well-executed AHU qualification in pharma ensures cleanroom compliance, prevents contamination, protects product quality, supports regulatory inspections, and maintains a controlled manufacturing environment. It is an essential part of HVAC validation and pharmaceutical quality assurance.





