














Operational Qualification of Purified Water System
Operational Qualification (OQ) of a purified water system is a documented verification activity performed to confirm that the system operates consistently within predefined operating limits. In pharmaceutical manufacturing, purified water is a critical utility used for formulation, cleaning, rinsing, and laboratory testing. Therefore, the purified water generation, storage, and distribution system must be qualified to ensure it delivers water of the required quality under routine operating conditions.
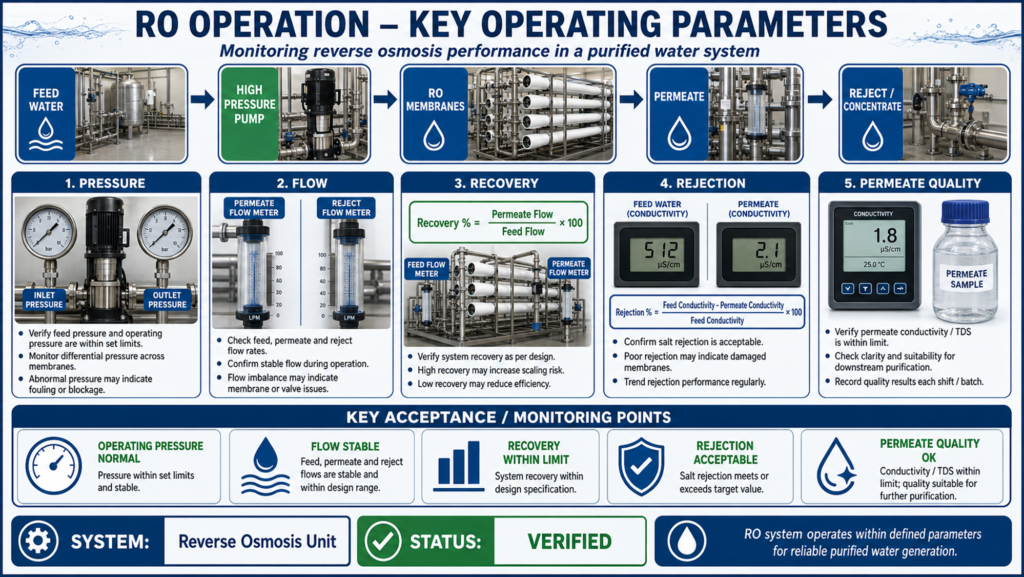
The OQ study is conducted after successful installation qualification. It verifies the functional performance of major components such as pumps, storage tank, distribution loop, valves, heat exchangers, filters, UV unit, instruments, control panels, alarms, and interlocks. Before starting OQ, the approved protocol, standard operating procedures, calibration status, utility availability, and system readiness are reviewed.
During operational qualification of purified water system, key parameters are challenged and monitored. These include flow rate, pressure, temperature, conductivity, total organic carbon, tank level, recirculation velocity, sanitization cycle, alarm response, and system recovery. Functional checks are performed to confirm pump operation, valve operation, loop circulation, automatic controls, emergency stop, low-level alarm, high-conductivity alarm, and other critical interlocks.
Sampling is an important part of OQ. Water samples are collected from the generation point, storage tank, return loop, and user points to verify chemical and microbiological quality. Test results must comply with approved acceptance criteria and applicable pharmacopoeial requirements.
All observations, printouts, trends, deviations, and corrective actions are recorded in the OQ report. Any failure or abnormal result must be investigated before final approval. A successfully completed OQ demonstrates that the purified water system is capable of operating as intended and is ready for performance qualification.
In conclusion, operational qualification of purified water system ensures regulatory compliance, process reliability, and consistent water quality. It helps pharmaceutical companies reduce contamination risk, maintain GMP standards, and support safe manufacturing operations.





