

















Performance Qualification of Purified Water System in Pharma
Performance Qualification (PQ) of a Purified Water System in pharmaceutical manufacturing is a critical validation activity performed to ensure the system consistently produces purified water meeting predefined quality standards during routine operation. In the pharmaceutical industry, purified water is widely used in manufacturing, cleaning, equipment washing, and laboratory applications. Therefore, maintaining water quality is essential for product safety, efficacy, and regulatory compliance.
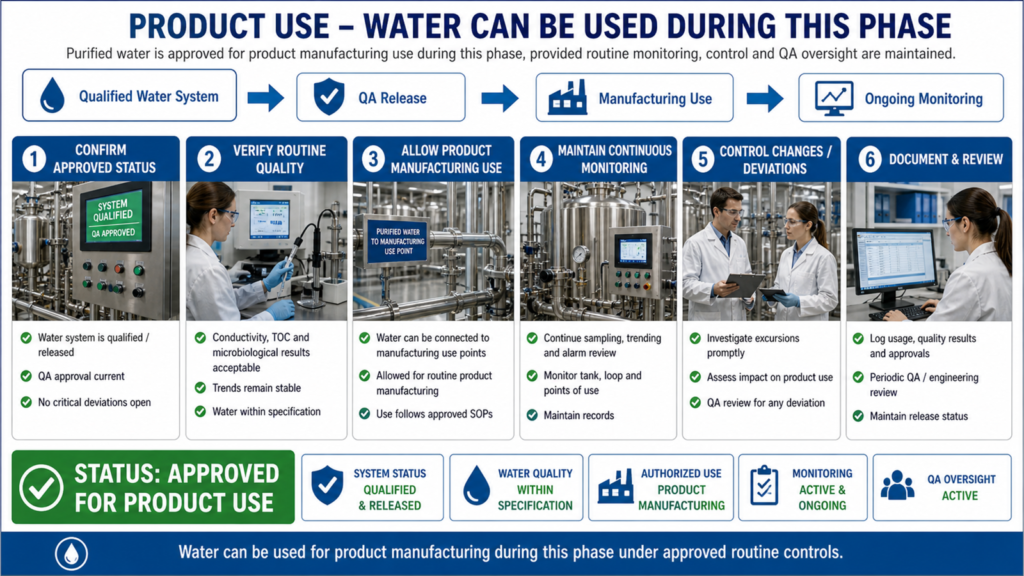
The Performance Qualification phase is conducted after successful completion of Design Qualification (DQ), Installation Qualification (IQ), and Operational Qualification (OQ). PQ verifies that the purified water system performs effectively under actual operating conditions over an extended period. During this phase, water samples are collected from various user points and storage locations to evaluate system consistency and reliability.
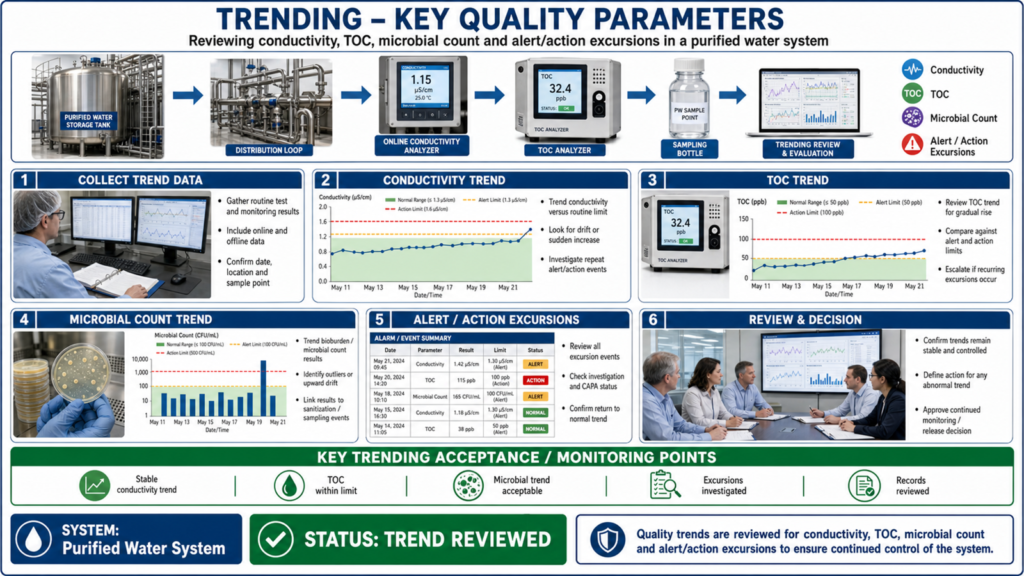
Key parameters monitored during Performance Qualification include conductivity, Total Organic Carbon (TOC), pH, microbial count, endotoxin levels, temperature, pressure, and flow rate. Testing frequency is usually intensive initially and gradually reduced after demonstrating stable system performance. Trending and documentation of results are essential to identify variations and ensure continuous compliance with pharmacopeial requirements such as USP, EP, IP, and WHO guidelines.
A typical purified water system includes pretreatment units, reverse osmosis (RO), electrodeionization (EDI), UV disinfection, storage tanks, and distribution loops. The PQ study confirms that all components work together efficiently to maintain purified water quality throughout the distribution system.
Proper documentation is an important part of Performance Qualification. Validation protocols, sampling plans, test reports, deviations, corrective actions, and final conclusions must be recorded accurately to satisfy GMP and regulatory audit requirements.
Effective Performance Qualification of a Purified Water System helps pharmaceutical companies minimize contamination risks, improve process reliability, maintain regulatory compliance, and ensure high-quality pharmaceutical product manufacturing. A well-qualified water system ultimately supports patient safety and overall product quality assurance in pharmaceutical operations.





