









Manufacturing Process of Acetylcysteine Effervescent Tablets USP 600 mg
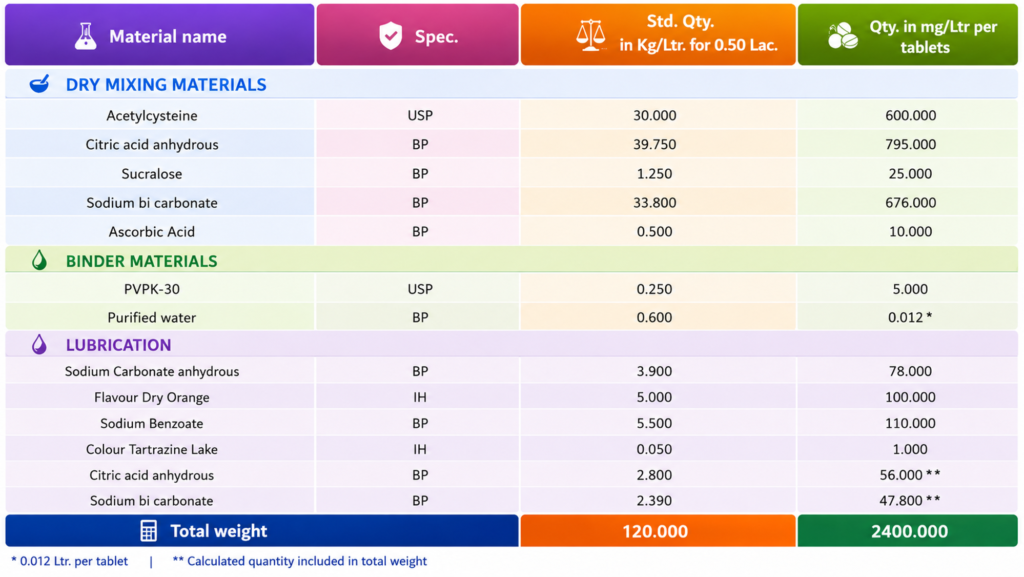
Manufacturing Acetylcysteine Effervescent Tablets USP 600 mg are manufactured under controlled low-humidity conditions because effervescent ingredients are highly moisture-sensitive. The process begins with dispensing of Acetylcysteine USP and excipients as per the approved master formula. Typical excipients include citric acid, tartaric acid, sodium bicarbonate, sodium carbonate, sweetener, flavour, binder, lubricant, and suitable filler.
All raw materials are checked for identity, assay, moisture content, and particle size. Materials are passed through a suitable sieve to remove lumps and ensure uniformity. Since acid and alkaline components react in the presence of moisture, they are usually processed separately or handled under relative humidity not more than about 25–30%.
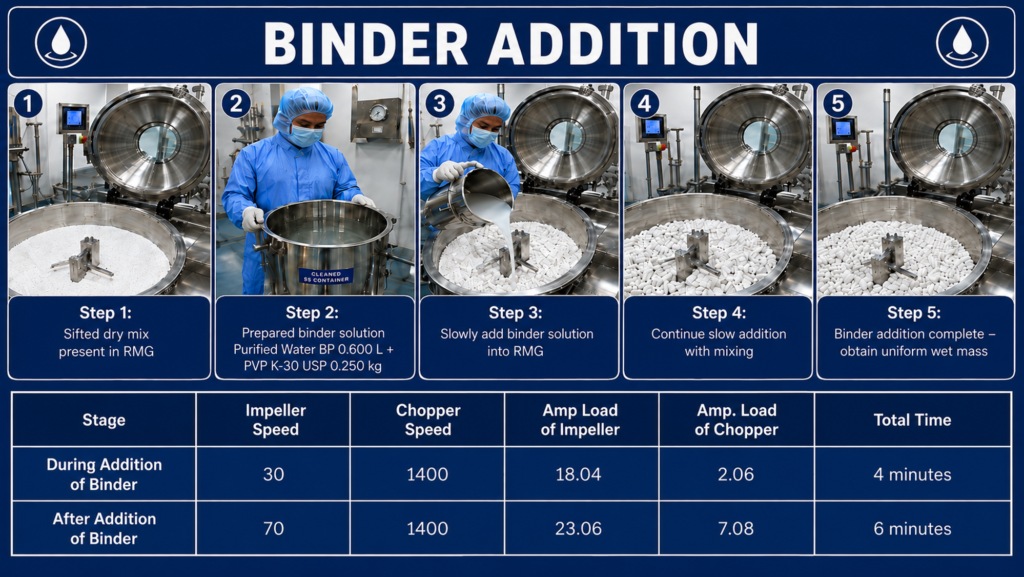
Acetylcysteine is blended with part of the acid component and filler to improve distribution. Separately, sodium bicarbonate and sodium carbonate are sifted and blended. If granulation is required, non-aqueous or dry granulation methods are preferred. Alcoholic binder solution or dry binder may be used to avoid premature effervescence. Granules are dried at controlled temperature, then milled and sifted to obtain uniform granule size.
The dried acid granules, alkaline granules, Acetylcysteine blend, flavour, sweetener, and other extra-granular excipients are mixed in a blender for a validated time. Lubricant such as sodium stearyl fumarate or polyethylene glycol is added at the final stage and blended gently to avoid over-lubrication.
The final blend is evaluated for appearance, bulk density, tapped density, flow property, moisture content, assay, and blend uniformity. Compression is performed using suitable tooling to produce large effervescent tablets with required hardness, thickness, weight, and friability. In-process checks include tablet weight variation, hardness, thickness, disintegration time, effervescence time, appearance, and assay.
After compression, tablets are immediately packed in moisture-protective packaging such as aluminium tubes with desiccant, strip packs, or alu-alu blisters. Finished tablets are tested for description, identification, assay, content uniformity, dissolution or disintegration, water content, effervescence time, microbial limits, and stability before release.





