



















A SOP for pest and rodent control in pharma facilities is a visual and practical guide designed to help personnel understand how to prevent, identify, monitor, and control pests effectively. In pharma industries, pest control is critical because insects, rodents, and other pests can contaminate raw materials, packaging components, finished products, and clean areas, leading to serious quality and regulatory issues.
The SOP should begin with the objective and scope, explaining that the purpose is to prevent pest infestation and maintain hygienic, GMP-compliant premises. It should visually show common pests found in pharmaceutical facilities, such as rats, mice, cockroaches, flies, ants, and stored-product insects. Clear pictures help staff recognize signs of infestation, including droppings, gnaw marks, damaged packaging, nests, foul odor, and insect presence.
The pictorial SOP should explain preventive measures. These include keeping doors closed, sealing cracks and gaps, maintaining proper drainage, using air curtains or door screens, storing materials neatly, removing waste regularly, and ensuring good housekeeping. Visual instructions should highlight that food is not allowed in production or storage areas and that spills must be cleaned immediately.
Monitoring activities should also be shown clearly. The SOP should include images of glue pads, rodent traps, bait stations, insect light traps, and monitoring maps. Each device should be placed at defined points and checked at specified intervals. The pictorial guide should also show how to record trap numbers, pest sightings, and inspection findings in a logbook or checklist.
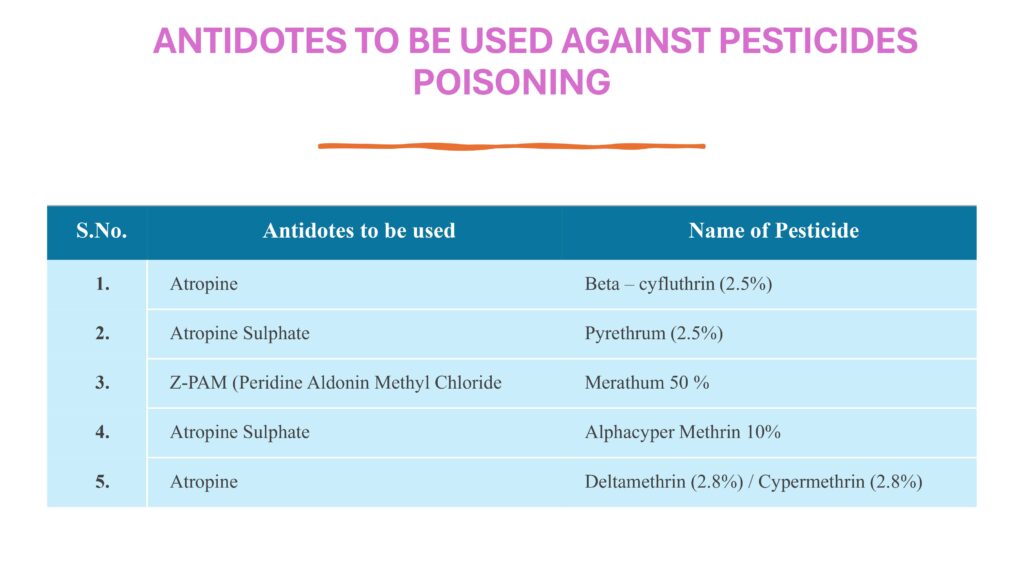
Corrective actions must be included. If pest activity is observed, the area should be inspected, affected materials assessed, housekeeping intensified, and the pest control vendor or responsible department informed immediately. The SOP should show that pesticides or rodenticides must only be used by authorized personnel and never in a way that risks contamination of products or equipment.
Finally, the pictorial SOP should emphasize documentation, trend review, and periodic evaluation. All inspections, actions taken, and observations must be recorded. A well-designed pictorial SOP improves awareness, ensures consistency, supports GMP compliance, and helps maintain a clean, safe, and pest-free pharmaceutical environment.





