














Purified Water Installation Qualification in Pharma
Purified Water Installation Qualification (IQ) is a documented verification process used in pharmaceutical industries to confirm that the purified water generation, storage, and distribution system is installed correctly as per approved design, user requirement specification, GMP guidelines, and engineering drawings. Purified water is widely used in manufacturing, cleaning, formulation, laboratory testing, and equipment washing; therefore, its system installation must be properly qualified before operational and performance qualification activities.
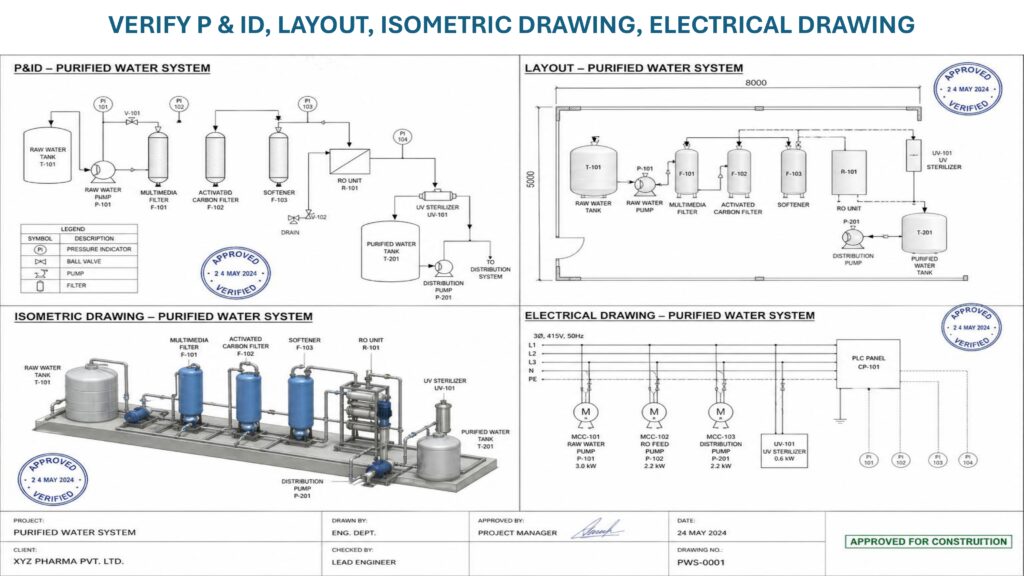
The main objective of Installation Qualification is to ensure that all components of the purified water system are installed as per approved specifications. These components may include raw water supply line, pre-treatment system, softener, activated carbon filter, micron filters, reverse osmosis unit, UV system, electro-deionization unit, storage tank, distribution loop, pumps, valves, instruments, sampling points, and piping.
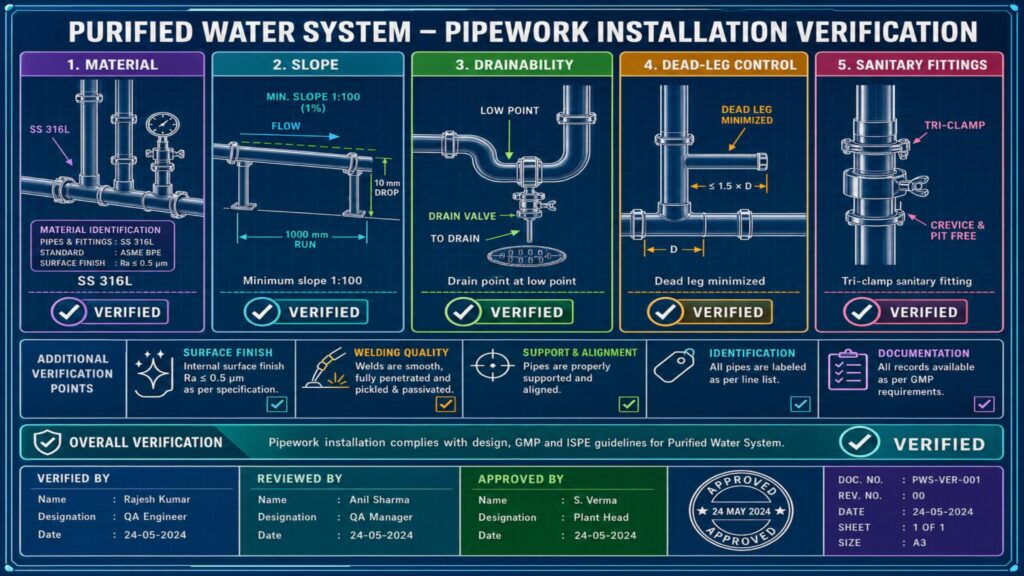
During IQ, documents such as purchase orders, equipment manuals, design qualification, P&ID, layout drawings, material certificates, welding records, calibration certificates, and installation checklists are verified. The installation team checks equipment identification, make, model, capacity, material of construction, pipe slope, drainability, dead legs, sanitary fittings, flow direction, utility connections, electrical connections, and safety requirements.
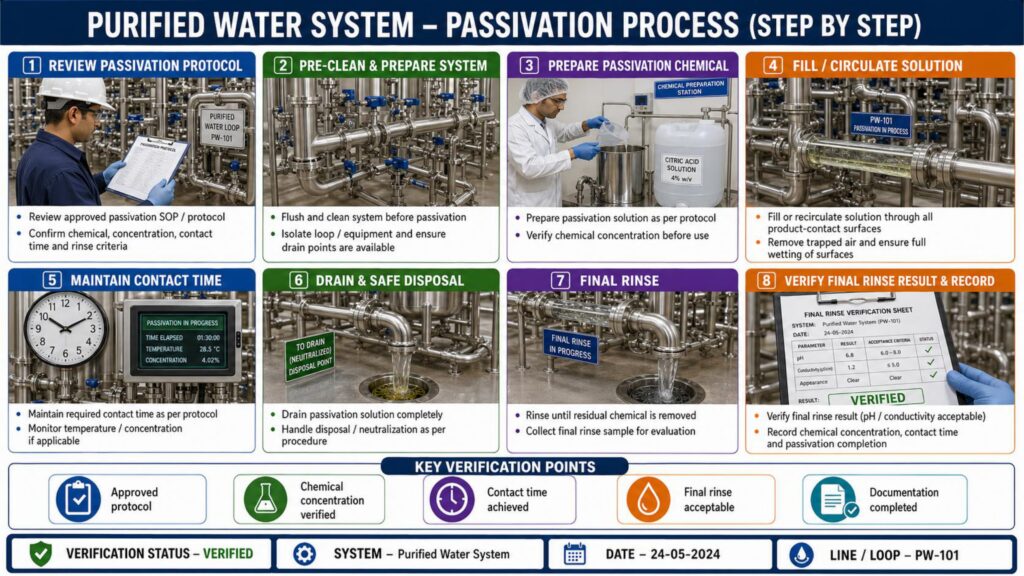
In pharma, purified water systems are generally constructed using stainless steel 316L with hygienic design to prevent microbial contamination and biofilm formation. Proper orbital welding, passivation, slope verification, and loop circulation are important aspects of qualification. All critical instruments such as pressure gauges, flow meters, temperature indicators, conductivity meters, and TOC analyzers must be installed and calibrated.
Any deviation observed during Installation Qualification must be recorded, investigated, and closed before proceeding to Operational Qualification (OQ). The final IQ report should summarize the installation verification results and provide approval from quality assurance, engineering, validation, and production departments.
A properly executed Purified Water Installation Qualification ensures regulatory compliance, system reliability, contamination control, and consistent water quality. It provides documented evidence that the purified water system is ready for further qualification and routine pharmaceutical use.





