

















An SOP for Cleaning & Sanitation of Premises in Pharma defines the standard procedure for maintaining cleanliness, hygiene, and contamination control in pharmaceutical manufacturing facilities. This SOP is essential for ensuring product quality, employee safety, regulatory compliance, and a clean GMP-compliant working environment.
The purpose of this SOP is to describe the systematic cleaning and sanitation procedure for production areas, packing areas, warehouses, laboratories, corridors, change rooms, washrooms, utility areas, and surrounding premises. Regular cleaning prevents dust accumulation, microbial contamination, pest attraction, cross-contamination, and product mix-up risks.
The SOP should clearly define the responsibilities of housekeeping staff, production personnel, supervisors, quality assurance, and department heads. Housekeeping staff are responsible for routine cleaning activities, while supervisors verify cleanliness. Quality assurance ensures that cleaning procedures, disinfectants, and records comply with GMP requirements.
Cleaning materials and equipment should include clean mops, lint-free cloths, scrubbers, vacuum cleaners, disinfectant solutions, detergents, buckets, floor wipers, waste bags, and approved PPE. Personnel must wear gloves, masks, aprons, and safety shoes before starting cleaning activities.
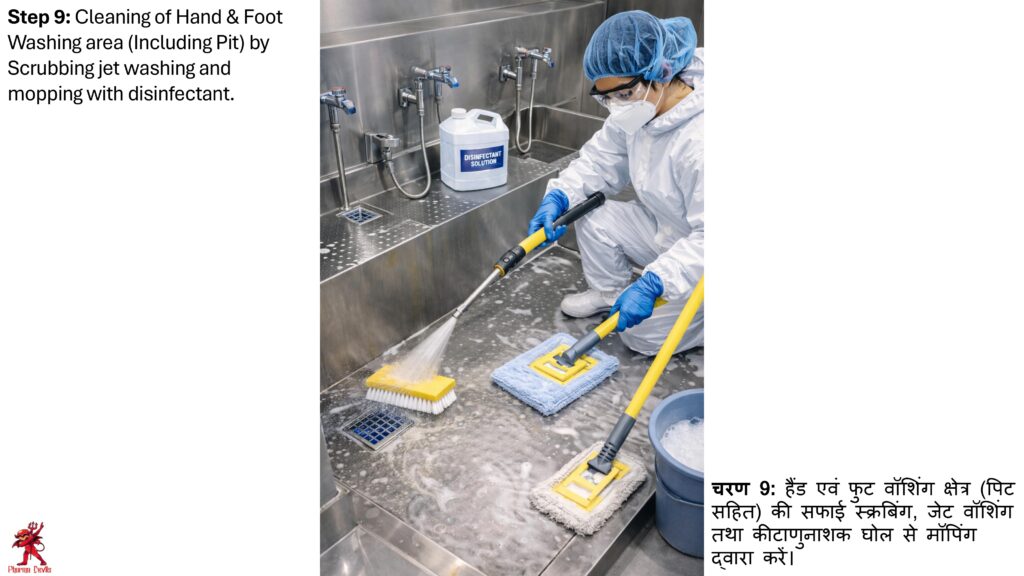
The cleaning procedure should be performed in a defined sequence. First, remove waste materials, unwanted items, and visible dirt from the area. Clean ceilings, walls, doors, windows, floors, drains, equipment exteriors, furniture, and fixtures as applicable. Dry cleaning, wet cleaning, vacuum cleaning, and mopping should be performed according to area classification and cleaning frequency.
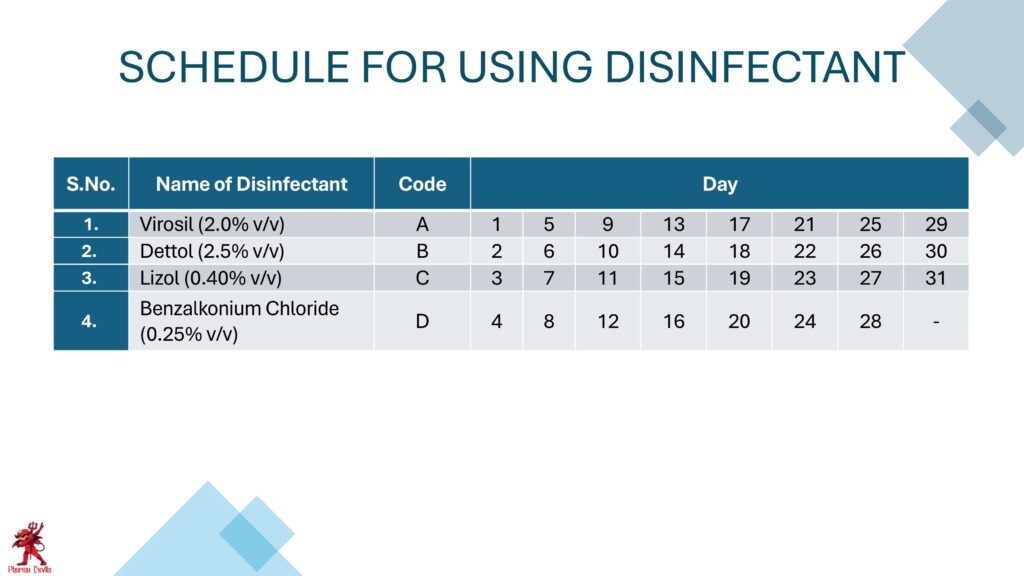
Sanitation should be carried out using approved disinfectants at defined concentration and contact time. Disinfectant rotation should be followed to avoid microbial resistance. Critical areas should be cleaned more frequently than general areas. Cleaning tools used in different areas should be color-coded and stored separately to prevent cross-contamination.
After cleaning, the area should be inspected for dust, stains, odor, spillages, waste, and water stagnation. Any abnormal observation must be reported and corrected immediately.
All cleaning and sanitation activities must be recorded in a logbook or checklist with date, time, area name, cleaning agent used, signature, and verification.
A well-implemented cleaning and sanitation SOP supports GMP compliance, prevents contamination, and maintains safe pharmaceutical premises.





