









An SOP for health checkup in pharma defines the standard procedure for medical examination and fitness evaluation of employees working in pharmaceutical facilities. The purpose of this SOP is to ensure that personnel are medically fit to perform their assigned duties and do not pose any contamination, safety, or health risk to products, processes, or other employees.
The SOP applies to all employees, contract workers, temporary staff, and personnel working in production, quality control, quality assurance, warehouse, engineering, packing, and other GMP-related areas. Health checkups should be conducted at the time of joining, periodically as per company policy, after long illness, after exposure to hazardous materials, and whenever required by management or regulatory expectations.
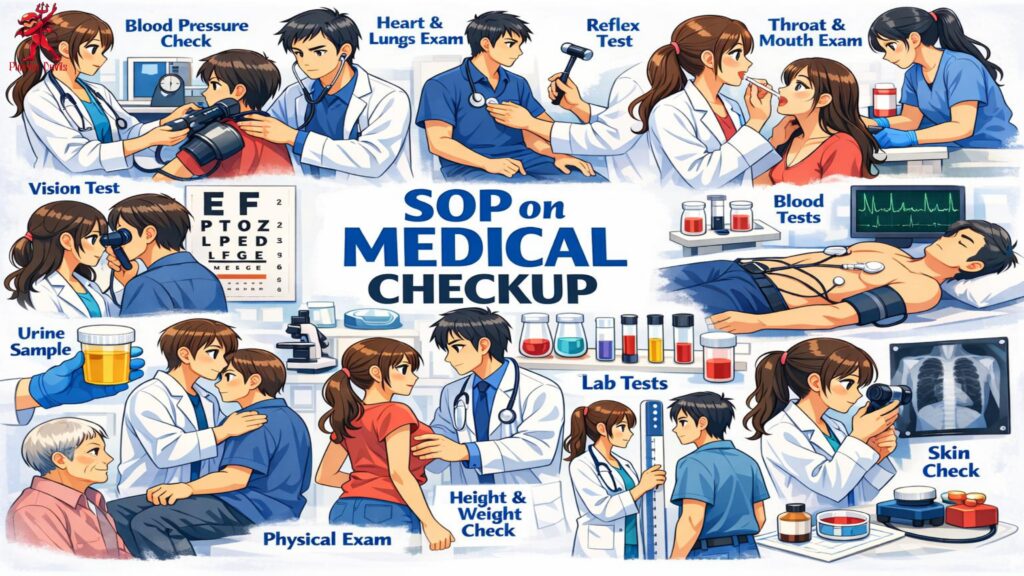
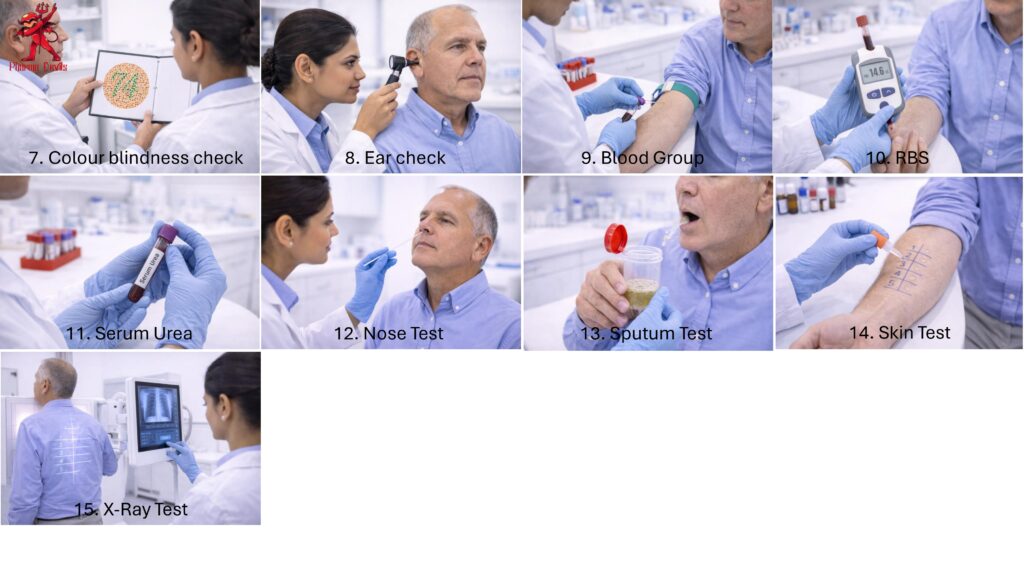
The procedure should include pre-employment medical examination, periodic health checkup, and special medical evaluation. The medical examination may include general physical examination, height, weight, blood pressure, pulse rate, vision test, hearing test, skin examination, respiratory assessment, blood tests, urine test, chest X-ray, and any job-specific medical tests. Employees working in sterile, potent, hormone, beta-lactam, or hazardous areas may require additional evaluation based on risk assessment.
The SOP should clearly define responsibilities. HR is responsible for coordinating health checkups and maintaining employee records. The medical officer or approved medical practitioner is responsible for conducting examinations and issuing fitness certificates. Department heads are responsible for ensuring that only medically fit employees are assigned to work in GMP areas.
If an employee is found suffering from communicable disease, open wound, skin infection, respiratory illness, or any condition that may affect product quality or employee safety, the person should be temporarily restricted from sensitive operations. Such cases should be reported to HR and the department head, and appropriate medical advice should be followed.
All health checkup records, fitness certificates, medical reports, and follow-up actions must be maintained confidentially. Records should include employee name, department, date of examination, medical findings, fitness status, doctor’s signature, and next due date.
A well-implemented SOP for health checkup in pharma supports GMP compliance, prevents contamination risk, protects employee health, and ensures a safe and quality-focused working environment.





