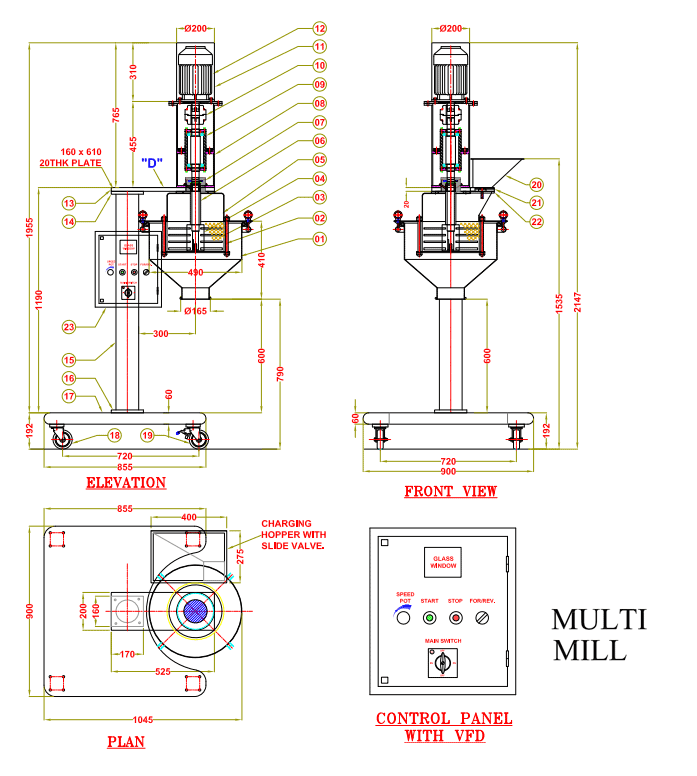
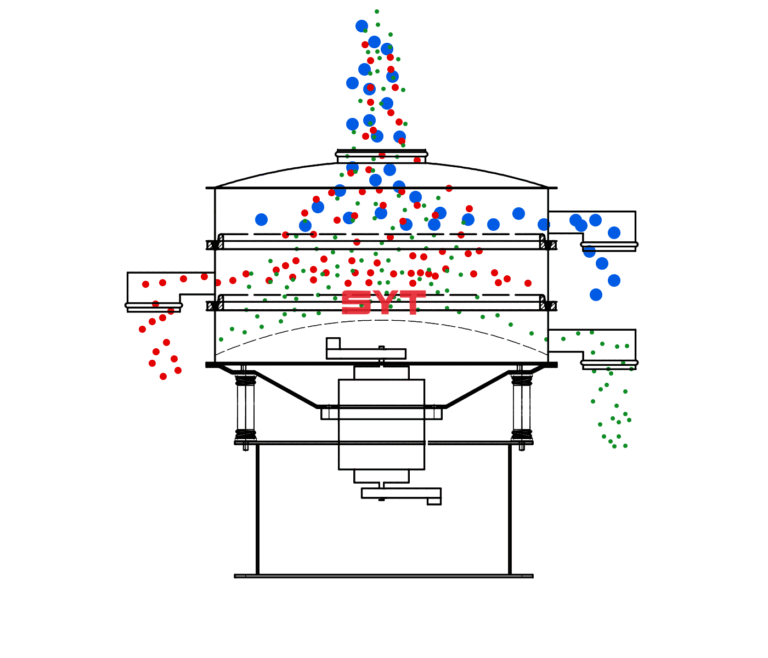
SOP FOR FORMULATIONS, RESEARCH AND DEVELOPMENT IN PHARMA

Pharmaceutical formulation and development involve transforming active pharmaceutical ingredients (API’s) into safe, effective, and patient-friendly medications. This process encompasses several key stages: 1. Pre-formulation Studies 2. Formulation Design 3. Process Development and Optimization 4. Quality by Design (QbD) 5. Regulatory…