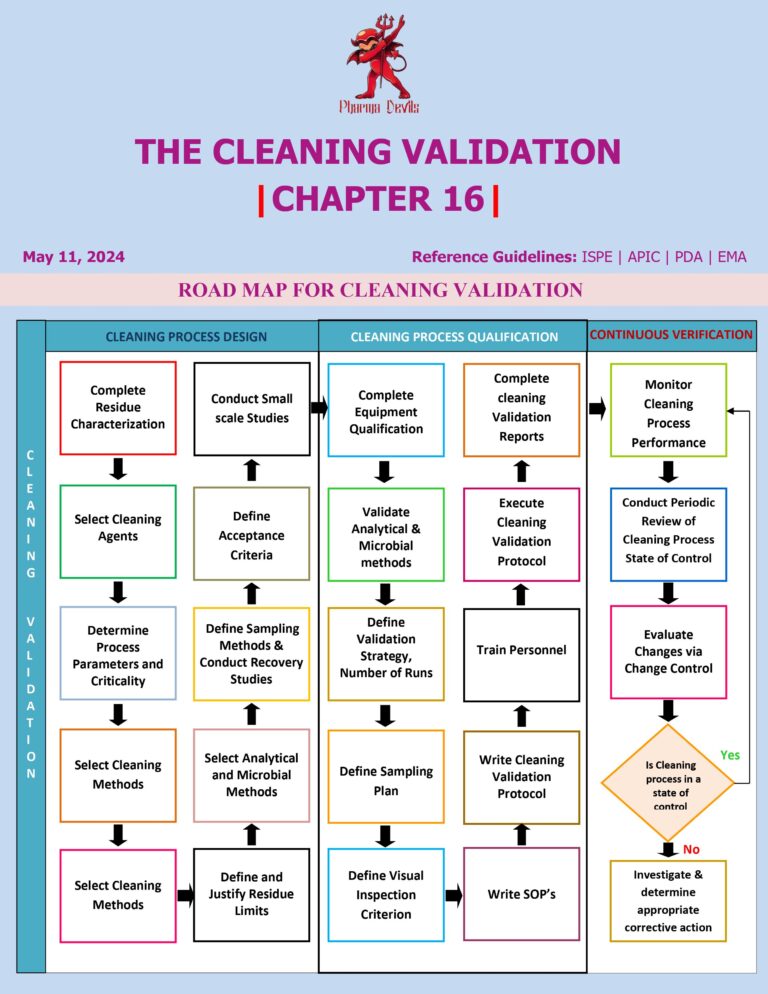
Cleaning Validation (Chapter 17) Establishing Process Parameters for a Manual Cleaning Process

Establishing process parameters for a manual cleaning process in cleaning validation means defining, controlling, and justifying the key variables that ensure consistent removal of product residues and cleaning agents from equipment. Because manual cleaning depends heavily on operator technique, parameters…